 With the increased deployment of vaccines against SARS-CoV-2, there is interest in determining vaccine efficacy and duration of immunity-related protection. Recent studies have shown a strong correlation between antibodies against SARS-CoV-2 in blood and saliva 15, 16, 17, 18. For example, good correlation has been reported between salivary and serum antibodies for human immunodeficiency virus (HIV) antibody testing 9, 12, 13, 14. There is a precedent for the utility of saliva for the detection of antibodies against various viral agents 9, 10, 11. 
It is a noninvasive alternative sample source that offers the flexibility of self-collection and home-based collection. Saliva is a clear mixture of extracellular secretions produced by salivary glands in the mouth that consists of approximately 99% water, while the remaining components consist of electrolytes, mucus, and protein including antimicrobial agents such as IgA and IgG 6, 7, 8. The majority of serological tests for antibodies against SARS-CoV-2 require a blood sample from an invasive venipuncture, which cannot be readily implemented in large population-based screening programs. Although serological assays are not intended for diagnostic purposes, antibody detection can be useful to evaluate the degree of immunization, measure seroprevalence, identify and trace contacts, and identify potential convalescent plasma donors 4, 5. The COVID-19 pandemic is unprecedented and continues to be a global public health emergency that has affected more than 230 million people worldwide, resulting in 4.7 million deaths as of September 2021 1, 2, 3. These findings support the suitability of concentrated saliva specimens for the measurement of SARS-CoV-2 RBD IgG antibodies via ELISA, and unconcentrated saliva specimens for the measurement of SARS-CoV-2 RBD IgG and IgA using an ultrasensitive Simoa immunoassay. An average relative increase of 431 pg/mL was observed when the unconcentrated saliva specimens were analyzed using the Simoa immunoassay (SARS-CoV-2 RBD IgG post/pre). When analyzed using the SARS-CoV-2 RBD IgG antibody ELISA, the average relative increase in antibody index (AI) between the saliva of the post- and pre-vaccinated individuals was 8.7 (AI post/pre). The diagnostic sensitivity of the saliva ELISA method was 90% (95% CI 76.3–97.2%) compared to 91.7% (95% CI 77.5–98.2%) for the Simoa immunoassay without total IgG-normalization and 100% (95% CI 90.3–100%) for the Simoa immunoassay after total IgG-normalization when compared to the serum ELISA assay. 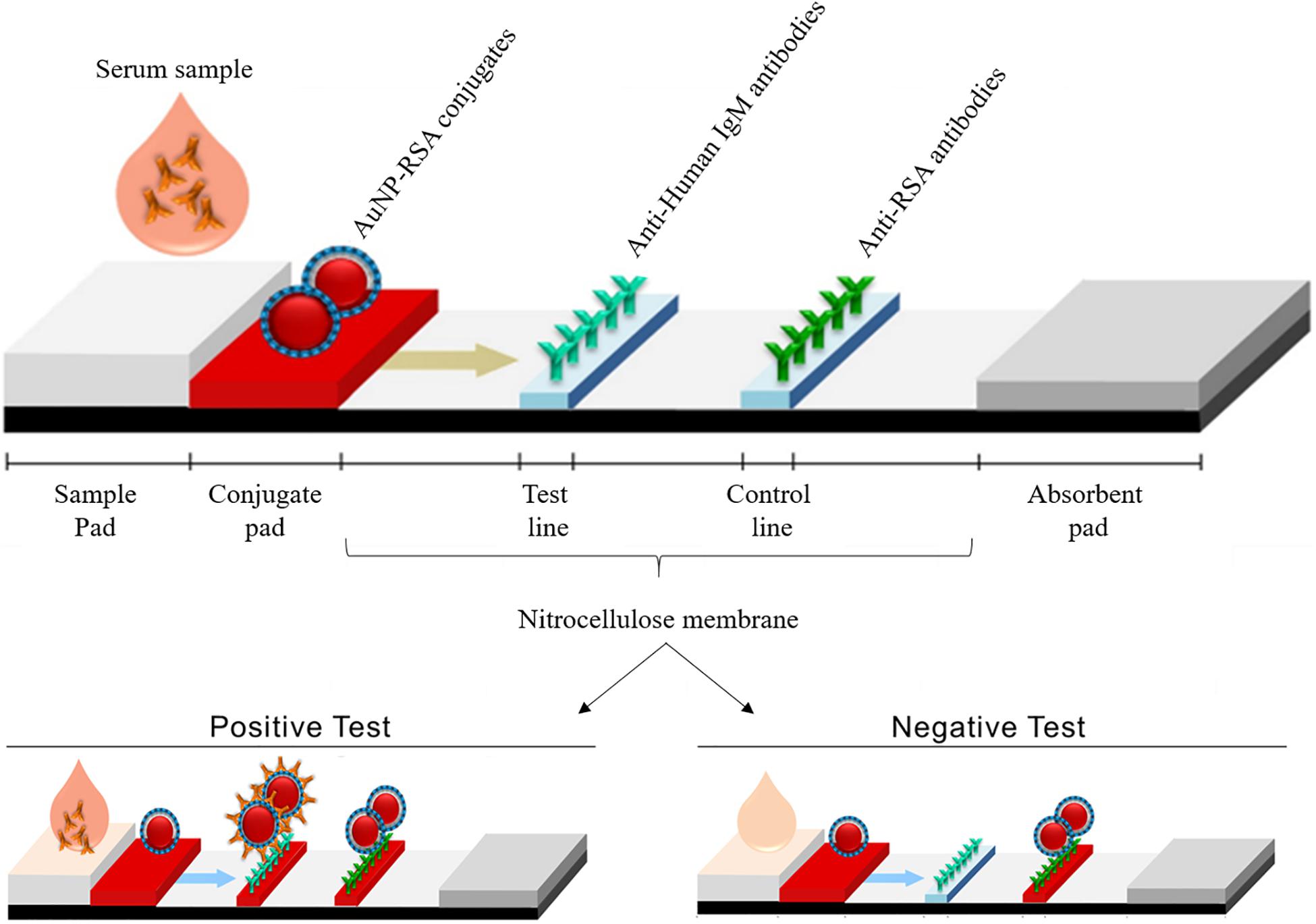
The analytical sensitivity of the saliva-based Simoa immunoassay was five orders of magnitude higher than the ELISA assay: 0.24 pg/mL compared to 15 ng/mL. 
The IgG results were compared with paired serum specimens that were analyzed for total RBD antibodies using the ELISA method. Saliva was collected using a commercially available collection device (OraSure Inc.) and SARS-CoV-2 RBD IgG antibodies were measured by an indirect ELISA using concentrated saliva samples and a Simoa immunoassay using unconcentrated saliva samples. Paired serum and saliva specimens were collected from individuals (n = 40) prior to and 2 weeks after receiving an initial prime COVID-19 vaccine dose (Pfizer/BioNTech BNT162b2 or Moderna mRNA-1273). The performance of the method was compared to a laboratory-developed SARS-CoV-2 RBD total antibody enzyme-linked immunosorbent assay (ELISA). We assessed the feasibility of a highly sensitive immunoassay method based on single molecule array (Simoa) technology to detect IgG and IgA antibodies against SARS-CoV-2 spike protein receptor binding domain (RBD) in saliva from individuals with natural or vaccine-induced COVID-19 immunity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
